Is Lanza’s Psychiatrist’s Drugging Indicative of Connecticut’s Mental Health Services?
Is Lanza’s Psychiatrist’s Drugging Indicative of Connecticut’s Mental Health Services?
In today’s Connecticut News Times there was a brief article spelling out the State’s legislative agenda, including efforts to block the right of the public to have access to information on homicides. This legislative measure is in response to the State’s continued unwillingness to make public the mental health records of Sandy Hook shooter, Adam Lanza.
The obvious point of having public disclosure of Lanza’s mental health records is two-fold. First, it is important to know whether Lanza was prescribed any psychiatric mind-altering drugs that may have played a part in his violent behavior. The disclosure of these records also is necessary in order for lawmakers to make informed decisions about costly, mental health legislation.
Ablechild has, for more than a year, used every legal means available in order to persuade the State to release Lanza’s mental health records. Apparently, the State is using Lanza as the backbone of enormous legislative efforts to increase mental health funding and services within the State, yet there is no evidence that Lanza used any State mental health services. All available information reveals that Lanza’s mental health was paid for by private insurance.
Based on the State Police Report, no mental health records are available for Lanza after 2007 when he was 15-years old. Additionally, Lanza’s apparent primary psychiatrist, Dr. Paul Fox, surrendered his license in June of 2012, destroyed his patient records and move to New Zealand, making it impossible to obtain Lanza’s mental health records from the primary source.
Whether Lanza had been prescribed psychiatric mind-altering drugs is extremely relevant to understanding his uncharacteristic violent behavior. Furthermore, it is important to remember that Connecticut Assistant Attorney General, Patrick B. Kwanashie, during the Ablechild FOIA hearing, revealed that the reason for not divulging the identity of the antidepressants he (Lanza) was taking was because it would “cause a lot of people to stop taking their medications.”
Based on this information, it certainly seems that Lanza had been prescribed, at a minimum, antidepressants. However, Ablechild has been contacted by a mother whose young son also had been a patient of Dr. Paul Fox and, based on this child’s medication record, one could easily question whether Lanza’s mental health “treatment” included just antidepressants.
For example, this unidentified ten-year old child began receiving “treatment” from Dr. Fox in 1998 for an apparent diagnosis of Asperger’s and, from what has been made public, Lanza also was diagnosed with Asperger’s at the age of five or six.
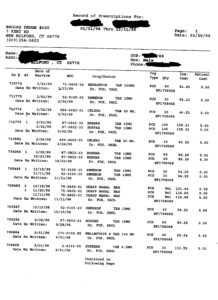
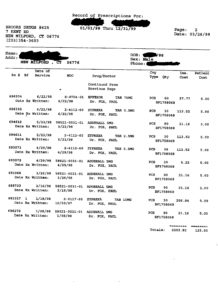
Between January 1998 and March of 1999 (14 months), Dr. Fox prescribed to the unidentified ten-year old the following antidepressants: Celexa, Remeron, Effexor and Wellbutrin. Additionally, Fox prescribed Neurontin, an Antiepileptic; Buspar, an Antianxiety; Zyprexa, an Antipsychotic; and Adderall, an Amphetamine.
As if prescribing eight mind-altering drugs to a ten-year old during a 14 month period isn’t outrageous enough, Dr. Fox prescribed these drugs as cocktails. The safety and efficacy for all of these drugs have not been established for pediatric use, nor are there any clinical trials showing safety and efficacy for pediatric use in any of these drug cocktail combinations. The majority of clinical trials last a few weeks or months. In other words, Dr. Fox doesn’t know how these drugs adversely affect a child’s developing brain when prescribed in combination, let alone singularly.
One daily drug regimen for the unidentified ten-year old includes the following:
8AM 1 Celexa (20mg)
4pm 1 Neurontin (300mg)
6PM 2 Buspar (15mg) each
8PM 1 Neurontin (300mg)
10PM 1 or 1/2 Remeron (15mg) if trouble sleeping.
Dr. Fox’s drug “treatment” regimen seems more in line with a chemical lobotomy, especially in light of the patient’s age. But there also is the added concern of adverse reactions associated with the mind-altering drugs prescribed.
For example, each of the Antidepressants carry the Food and Drug Administration’s (FDA) Black Box Warning. This is the federal agency’s most serious warning, which includes increased suicidal thinking and suicidality. More importantly, all of the Antidepressant drugs listed report adverse reactions such as “mood and behavior changes, anxiety, agitated, hostile, aggressive and hallucinations” to name a few.
Recall that Adam Lanza received the same diagnosis as our unidentified ten-year old. Is it possible that Dr. Fox chose a similar, or worse, drug “treatment” regimen for Adam Lanza? Who knows? The State of Connecticut apparently doesn’t want the public to know and is doing everything legally and legislatively possible to block access to Lanza’s mental health records.
After reading the list of mind-altering drugs prescribed to our ten-year old unidentified child, the State should be asking Dr. Fox some serious questions. And rather than spending so much time and effort trying to convince the people of Connecticut that increased mental health (which equates to increased drugging) is the answer to the tragedy at Sandy Hook, wouldn’t it be more beneficial to investigate the outrageous psychiatric drugging of the state’s children?
If Dr. Fox’s drug “treatment” regimen is indicative of the type of mental health being provided by the State of Connecticut, increasing services does not bode well for its children.